makes a resurgence in breast reconstruction
Plastic surgeons have these goals when they perform breast reconstruction:
• to accurately reconstruct landmarks of the breast (such as the location of the inframammary fold);
• to restore breast projection; and
• to provide a natural, mildly ptotic breast shape, which, above all else, is symmetric with the opposite side.
The earliest attempts at breast reconstruction used tissue from the opposite breast and the abdomen that was carried on tubed pedicles to cover the mastectomy site. While ingenious, these techniques tended to provide rather poor aesthetic results.
The possibilities for more aesthetic results improved significantly in 1963, when Cronin and Gerow introduced the silicone gel prosthesis for delayed reconstruction,1 and this became the favored approach throughout the 1960s and the early 1970s. In 1972, Radovan introduced the concept of tissue expansion and successfully applied it to breast reconstruction.2 As improvements in expander and breast implant design were made, this technique came to enjoy the widespread popularity that still persists today.
In the late 1970s, the latissimus dorsi (LD) musculocutaneous flap was rediscovered as a useful flap for breast reconstruction.3 (It was first described in 1906.) In 1982, however, Hartrampf et al described and subsequently popularized the use of the transverse rectus abdominis musculocutaneous (TRAM) flap for breast reconstruction.4 The TRAM flap has since become the “gold standard” by which all other methods of breast reconstruction are judged, and made the LD flap a rarely used option for many surgeons.
Recently, however, the LD flap has regained a measure of popularity, in part because of significant improvements in tissue expander and breast implant design. With strategic planning and optimal use of anatomic relationships, long-lasting results have been obtained using the LD flap—along with tissue expanders and implants—that are every bit equal to those obtained using the TRAM flap. In addition, using the LD flap avoids the potential morbidity associated with the harvest of the rectus abdominis muscle.
This surgical strategy is called the volume-added latissimus dorsi flap technique. It consistently provides adequate skin coverage and an aesthetic reconstruction of the breast-mound contour, resulting in im-proved breast symmetry and outstanding aesthetic results.
Anatomy
The latissimus dorsi muscle is the largest muscle in the body, and it is useful for covering extremely large wounds. Despite its size, no practical functional motor deficit results from its transposition. The muscle originates on the iliac crest inferiorly and the thoracolumbar fascia posteriorly near the midline. It inserts into the humerus, where it acts as a humeral adductor and internal rotator. The posterior axillary fold is comprised of the most superior aspect of the muscle that begins to narrow before it forms the tendon of insertion. The nerve supply is via the thoracodorsal nerve, a branch of the posterior cord of the brachial plexus.
Blood is supplied to the latissimus dorsi muscle via the subscapular artery, a branch of the axillary artery. The subscapular sends off a circumflex scapular branch posteriorly, then distributes a serratus branch before it enters the substance of the muscle on its undersurface as the thoracodorsal artery. A 5–10-cm pedicle can be obtained off the subscapular system. A single venae comitantes typically accompanies the artery.
The pedicle can be approached directly by dissecting the latissimus dorsi from the axilla, or it can be found by following the undersurface of the muscle in a distal to proximal approach. Because the artery divides in the substance of the muscle, the muscle can be split longitudinally to form a bilobed, or two-tongued, flap. The muscle also is supplied by perforators from the thoracic intercostal and lumbar arteries that allow it to be used as a pedicled flap that can resurface posterior defects.

Indications and Planning
Because of the robust vascularity present, the volume-added LD flap can be safely elevated in patients who are heavy smokers, diabetics, or obese, or have a pre-existing medical condition (such as hypertension) that places them at risk for ischemia. In addition, patients who have inadequate abdominal tissue for a TRAM flap, have had a previous TRAM flap excision on the other side, or prefer the scar on the back rather than on the abdomen (as with loss of the rectus abdominis muscle), are excellent candidates for the volume-added LD flap.
Accurate preoperative markings are vital for proper positioning of the skin island, and they should always be made with the patient upright. The flap’s superior margin is identified by locating the tip of the scapula and drawing a line from that landmark to the posterior axillary fold. The flap’s lateral margin is marked by drawing a straight line from the posterior axillary fold to the iliac crest. The medial border is the posterior midline, and the inferior border is the iliac crest.

Surgical Technique
Unilateral and bilateral flap elevations are both done with the patient prone. The skin is incised, and the deep layer of fat under the deep dorsal fascia of the back is harvested with the flap to add bulk and, eventually, to soften the peripheral contours of the mastectomy defect. This fascia is easily identified once the skin incision is made and the plane of dissection becomes obvious, particularly when adequate traction or countertraction is applied to the wound.
The superficial layer of fat destined to remain attached to the back flaps has a firm consistency and lies above the deep fascial layer. The deep layer of fat, harvested with the muscle, has a looser consistency and dissects easily from the underside of the fascial layer. Dissection extends through the deep layer of fat at the muscle margins, where the superior, medial, and inferior muscle attachments are released.
|
|
The anterior muscle border is dissected free; care should be taken not to inadvertently elevate the serratus anterior muscle. Dissection continues up into the posterior axilla. Once the flap is elevated, a communicating tunnel to the mastectomy defect is made high in the axilla and the flap is transposed anteriorly (Figure 2). A quilting technique is used to close the back incision to minimize seroma formation, and a drain is inserted.
The patient is rotated into the supine position, the entire chest is prepped and draped, and the mastectomy wound is evaluated. In the immediate setting, the surgeon must be convinced that the mastectomy flaps have adequate vascularity to survive. Debridement of any portion of the mastectomy flaps threatened by ischemia is performed as needed. The entire LD flap is pulled into the mastectomy defect, and the fibers of insertion are released high in the axilla.
Approximately 90% of the insertion is released, giving the flap up to 10 extra centimeters of excursion onto the chest wall. This allows the skin island to be rotated into any desired position. By keeping 10% of the insertion intact, inadvertent traction on the pedicle with potential stretch injury is prevented. The thoracodorsal nerve is also divided to prevent unwanted animation of the breast after the surgery. It has not been our observation that dividing the nerve affects the subsequent volume of the breast in any meaningful way.
Once transposed anteriorly, the edges of the LD flap are inset medially, superiorly, and laterally into the peripheral margins of the mastectomy defect. The bulk of the flap with the combined muscle and fat is designed to soften these contours and create a smooth takeoff from the chest wall onto the reconstructed breast.
Whether immediate or delayed, the skin island is inset where needed to create the most aesthetic shape possible. In immediate cases, this usually involves a small, circular skin island designed to replace a missing nipple and areola. In delayed cases, orienting the elliptical skin island low and lateral provides the best shape.
The tissue expander is inserted, and the flap is inset along the inframammary fold. We prefer to use a shaped textured tissue expander that has two small tabs attached at the inferomedial and inferolateral corners. The tabs can be sutured to the chest wall to hold the device in position while the LD flap is sutured around it. This maneuver can easily be performed with the patient upright, because the expander cannot shift its position. This design feature improves the accuracy of expander placement.
Very often, 200–300 mL of saline solution is placed in the expander at the time of surgery, and no postoperative inflation of the device is needed. Finally, the skin island is inset, and drains are placed (Figures 3, 4, and 5).
The procedure requires 3–6 hours for surgery and a 2–3-day hospital stay. Full return to activity occurs within 2–4 weeks. After surgery, the expander is filled as needed to increase the volume of the breast. After 6 months, the expander is removed and replaced with a permanent implant, and the nipple and areola are reconstructed. Any breast revision required to provide the best result possible is also performed, including capsulectomy, skin resection, and liposuction, as needed.

|
Results
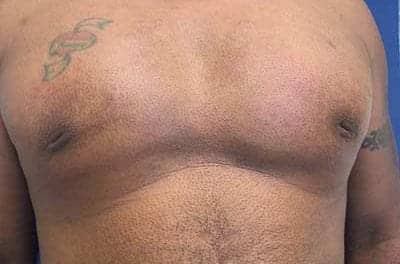
The volume-added LD flap technique is a versatile, predictable, and widely applicable technique for both immediate and delayed breast reconstruction. In patients who require only a small circular skin island after a periareolar skin-sparing mastectomy, the technique is particularly advantageous because the LD flap has a robust vascular supply and few donor-site complications (Figure 6). Bilateral cases are done as readily as unilateral cases, with a minimal increase in morbidity (Figure 7).
The technique also is an excellent salvage procedure for patients who have experienced complications after initial attempts at breast reconstruction were performed. The reliable vascular supply to the LD flap and the ability to provide a significant amount of volume to the breast with the tissue expander–implant combination provide versatility in managing a wide variety of reconstructive problems.
Surgery performed in a predictable and consistent fashion is particularly advantageous for patients who have already experienced complications (Figure 8). It should be noted, however, that the subset of patients who require a large skin paddle to resurface the breast mound adequately—as in cases of skin injury after radiation therapy—are sometimes better served with a TRAM flap, in which more skin is readily provided by the flap.
Complications
Vascular compromise to the LD is rare. In smokers, breakdown of the back donor incision has been noted occasionally, and simple secondary closure of the dehiscence is a very straightforward remedy. Capsular contracture is a risk, as it is with any implant procedure, but the additional soft tissue surrounding the device tends to soften the effects of the contracture. With severe contracture, traditional capsulectomy with implant replacement is easily accomplished.
The most common surgical sequelae of raising a LD flap is seroma formation in the back donor site. At times, the drains are left in place for up to 6 weeks after surgery, with intermittent percutaneous drainage performed once per week until it ceases. The use of quilting sutures in the closure of the back donor site has been useful for decreasing the need for prolonged drainage of the back.
In summary, the volume-added LD musculocutaneous flap has been demonstrated to be a safe and reliable method of breast reconstruction with minimal complications. When used with the newer implants and tissue expanders that are now available, it can achieve superior aesthetic results.
Dana Khuthaila, MD, is a Canadian Certified Plastic Surgeon and a fellow at the Center for Breast and Body Contouring in Grand Rapids, Mich. Dennis Hammond, MD, founded the center. He has been in private practice in Grand Rapids for 13 years, after having completed a 1-year fellowship with Pat Maxwell, MD, and Jack Fisher, MD, in Nashville, Tenn. The authors can be reached at (616) 464-4420 or via the center’s Web site, www.dennischammond.com.
References
1. Cronin TD, Gerow FJ. Augmentation mammaplasty: A new ‘natural feel’ prosthesis. Proceedings of the 11th Congress of Plastic Surgery, Washington DC. Excerpta Medica International Congress Series Number 66. Amsterdam: Excerpta Medica Foundation; 1963;41–49.
2. Radovan, C. Tissue expansion in soft-tissue reconstruction. Br J Plast Surg. 1972;25:37.
3. Schneider WJ, Hill HL, Brown RG. Latissimus dorsi myocutaneous flap for breast reconstruction. Br J Plast Surg. 1977;30:277.
4. Hartrampf CR, Scheflan M, Black PW. Breast reconstruction with a transverse abdominal island flap. Plast Reconstr Surg. 1982;69:216.