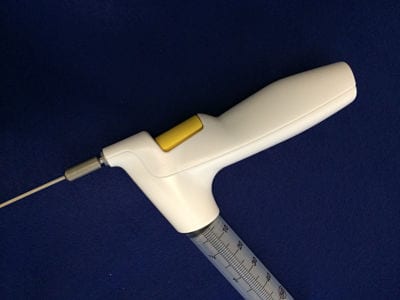
Fat grafting is back and it’s here to stay, but its widespread adoption has been mired by the lack of predictabilite results. The TauTona Tissue Injector (TTI) may help solve this and other problems associated with fat grafting.
The US Food and Drug Administration just cleared the single-use, handheld adipose tissue transfer tool for marketing
“Today most fat grafting is done using a 100-year-old technology: the syringe – a device that can be
clumsy for clinicians to use,” says Geoffrey Gurtner, MD, managing partner at TauTona and professor of plastic and reconstructive surgery at the Stanford School of Medicine, in a news release. “Syringes often clog and can lead to difficulty in tissue transfer procedures. The TauTona Tissue Injector is designed to help physicians simplify their fat grafting procedures, which may lead to reduced costs.”
The TTI is a battery powered, single-use tool that is designed to deliver adipose tissue at an even, controlled rate, allowing the surgeon to focus on placement of the fat. It is compatible with
off-the-shelf cannulas, syringes (reservoir), and current fat processing techniques.
“The absence of a specialized fat grafting tool has made it more difficult for surgeons to provide their patients with precise, repeatable results,” Gurtner says. “We believe that an effective fat transfer tool could increase the number of physicians using the technique and increase the number of procedures performed.”
The company also announced that it has received a key US Patent for TTI from the US Patent and Trademark Office (USPTO) and that it has other patents pending for the tool.