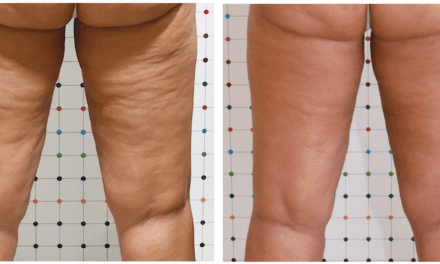
Sentient Launches Cellulite Reduction Device in U.S.
Sentient has launched Sentient Sculpt, a non-invasive cellulite reduction device that uses electromagnetic wave and microwave technology to target cellulite and fat cells while cooling the skin, with successful clinical results.