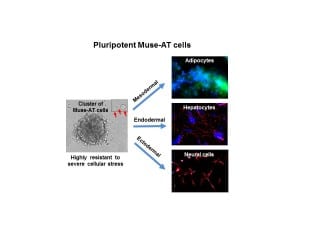
Researchers have isolated a new population of primitive, stress-resistant human pluripotent stem cells easily derived from fat tissue that are able to differentiate into virtually every cell type in the human body without genetic modification.
 |
Multi-lineage Stress-Enduring (Muse-AT) stem cells from adipose tissue were discovered by “scientific accident” when a piece of equipment failed in the lab, killing all the stem cells in the experiment except for the Muse-AT cells. Researchers from the UCLA Department of Obstetrics and Gynecology further discovered that not only are Muse-AT cells able to survive severe stress, they may even be activated by it.
These pluripotent cells, which are isolated from fat tissue removed during liposuction, expressed many embryonic stem cell markers and were able to differentiate into muscle, bone, fat, cardiac, neuronal and liver cells. An examination of their genetic characteristics confirmed their specialized functions, as well as their capacity to regenerate tissue when transplanted back into the body following their “awakening.”
“This population of cells lies dormant in the fat tissue until it is subjected to very harsh conditions. These cells can survive in conditions in which usually only cancer cells can live,” says senior author Gregorio Chazenbalk, PhD, an associate researcher with UCLA Department of Obstetrics and Gynecology and a scientist with the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research, in a news release. “Upon further investigation and clinical trials, these cells could prove a revolutionary treatment option for numerous diseases, including heart disease, stroke and for tissue damage and neural regeneration.”
The results of the 2-year study are published June 5, 2013 in the peer-reviewed journal PLOS ONE.
Purifying and isolating Muse-AT cells does not require the use of a cell sorter or other devices. They are able to grow either in suspension, forming cell spheres, or as adherent cells, forming cell aggregates very similar to human embryonic stem cell-derived embryoid bodies, the researchers write.
“We have been able to isolate these cells using a simple and efficient method that takes about 6 hours from the time the fat tissue is harvested,” Chazenbalk explains. “This research offers a new and exciting source of fat stem cells with pluripotent characteristics, as well as a new method for quickly isolating them. These cells also appear to be more primitive than the average fat stem cells, making them potentially superior sources for regenerative medicine.”
Going forward, Chazenbalk and colleagues will use Muse-AT cells in animal models to regenerate damaged or dysfunctional tissue to determine how efficiently they grow and perform in the body and to gauge their potential for future clinical use.
“Because lipoaspiration is a safe and noninvasive procedure and Muse-AT cell isolation requires a simple yet highly efficient purification technique, Muse-AT cells could provide an ideal source of pluripotent-like stem cells,” the researchers write. “Muse-AT cells have the potential to make a critical impact on the field of regenerative medicine.”